Results
Seasonal differences in water chemistry
June samples showed no substantial difference in mercury and methylmercury concentrations between the inflow and the outflow, and overall lower methylmercury concentrations at the inflow than August samples (Figure 10). In August, observed a mean 37.9% reduction in dissolved methylmercury, and we are 98% certain that the true change is a decrease (Figure 10).
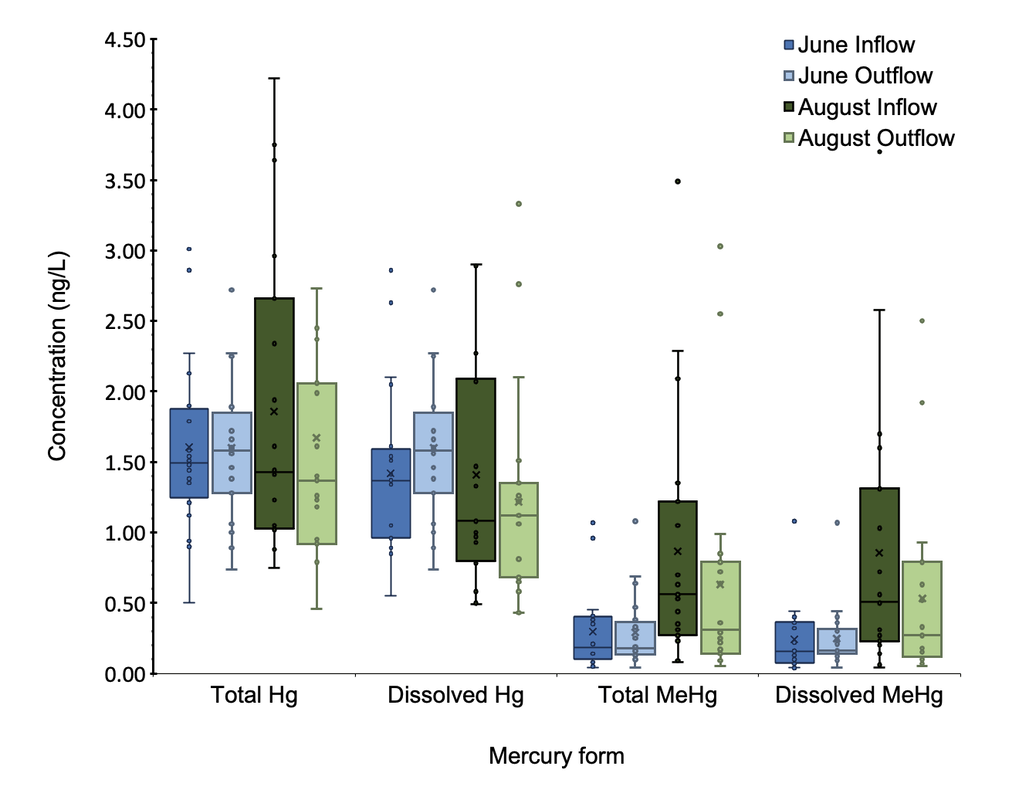
Figure 10. Boxplots of mercury and methylmercury concentrations for the inflow and outflow of June and August. One-sided T-tests indicate that in June, there is no significant change of MeHg concentration from inflow to outflow. In August, dissolved methylmercury concentration decreased an average of 37.9% (p=0.0198).
|
The change in methylmercury at the inflow between June and August may be explained by origin of water entering the pond, and the flow rate. Springtime (June) is characterized by high flow from snowmelt, causing an influx of low salinity water with lower residence time (Figure 11b). With less time for chemical transformations to occur within the pond, the water is chemically similar at the inflow and the outflow, especially when compared to low flow seasons (August).
Water entering the system in August is discharged from peatlands, so has more dissolved metals and ions when compared to snowmelt water in June (Figure 11.b). Peat water has higher methylmercury levels than fresh snowmelt water which results in a lower inflow methylmercury concentration in June. Ultimately, June and August water chemistry respond differently to pond conditions (Figure 11.a). As June showed no significant change in Methylmercury concentration, I will use August data only to explore relationships between other water chemistry variables. |
Figure 11. Water chemistry differences between June and August.
Figure 11.a. PCA ordination of seasonal changes to water chemistry. Ordination and points represent values from June samples. Arrows show the change in seasonality based on the August sample of the same sites. Figure 11.b. Box plot of Salinity and Residence time for June and August. Residence time is based off of the -Δ LCXS, so longer residence times are at the top of the boxplot. |
Methylmercury concentration change relative to other water chemistry variables
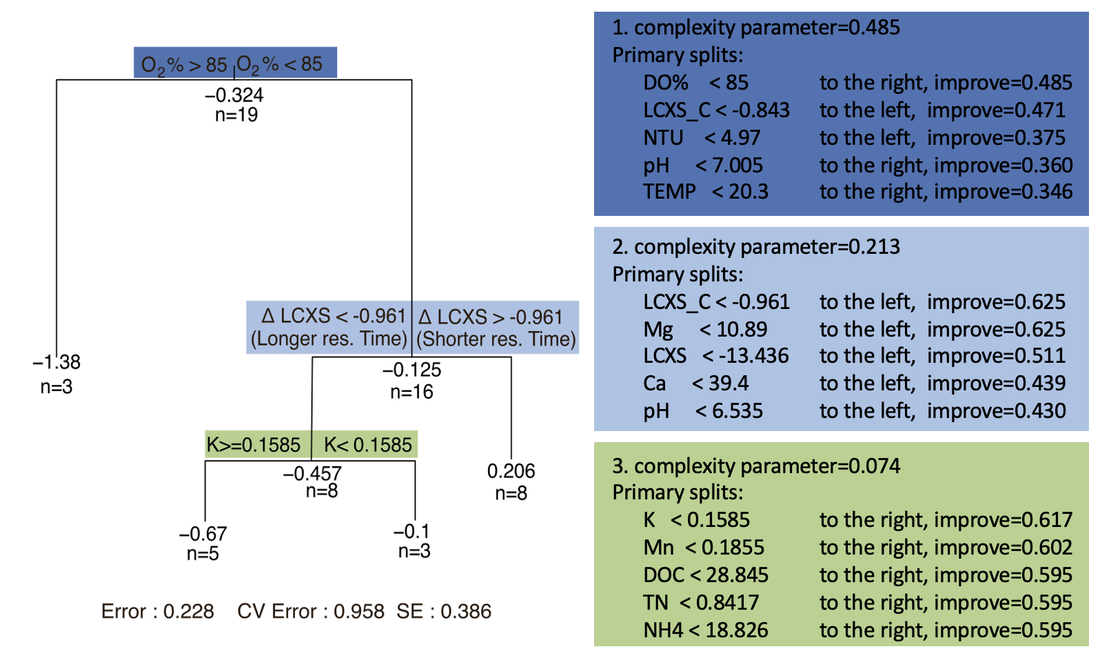
Methylmercury concentration changes are correlated with residence time, and dissolved oxygen (Figure 12). There are greater methylmercury decreases in ponds with higher oxygen levels, and longer residence times (Figure 12, Figure 13).
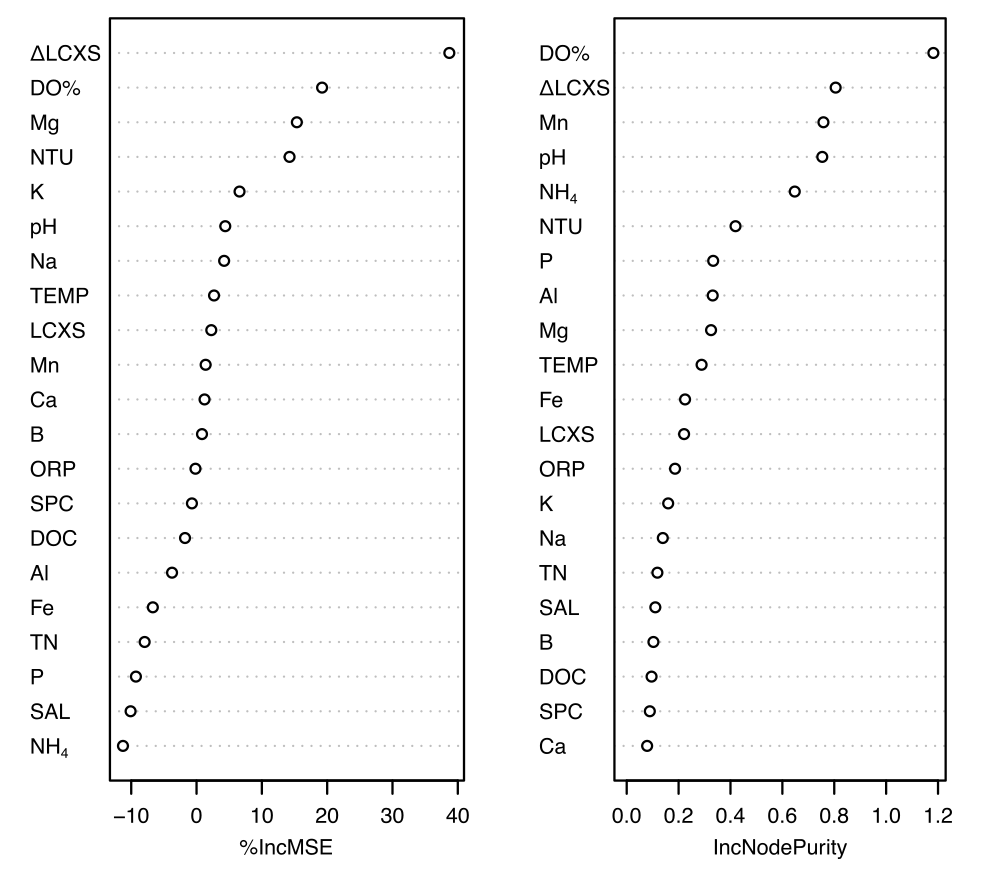
A classification and regression tree of methylmercury change shows similar results where the first two nodes which make up 69.8% of the variance are driven by dissolved oxygen, and relative residence time (Figure 14). After residence time and dissolved oxygen levels, redox-sensitive measurements NH4, Mn, and Mg have high importance for the model suggesting that the methylmercury changes are closely related to the redox conditions in the pond (Figure 15). Interestingly, dissolved carbon is a relatively unimportant variable to the model (Figure 15), and has a poor relationship with methylmercury changes (Figure 13) despite being a critical component in methylmercury transformations. To effectively evaluate carbon influence, I need to further explore the type and lability of carbon present. Regardless, methylmercury concentration decrease and the importance of residence time and redox conditions indicates that demethylation is occurring in the ponds.
Demethylation can occur through two primary pathways: Microbial demethylation, and photo demethylation (Lehnherr, 2014). Microbial demethylation occurs when microbes break down methylmercury using free oxygen in the water producing mercury, CO2, and CH4. Oxygen level correlation with methylmercury changes in the beaver ponds suggests that microbial demethylation is occurring.
Photo demethylation occurs when solar energy decomposes methylmercury in surface waters (Lehnherr, 2014). With longer residence time water is exposed to sunlight for a longer period of time, improving the likelihood of photolytic demethylation. Ponds with high methylmercury concentration changes also tend to have low turbidity (NTU) (Figure 13), increasing light penetration to deeper parts of the pond.
Photo demethylation occurs when solar energy decomposes methylmercury in surface waters (Lehnherr, 2014). With longer residence time water is exposed to sunlight for a longer period of time, improving the likelihood of photolytic demethylation. Ponds with high methylmercury concentration changes also tend to have low turbidity (NTU) (Figure 13), increasing light penetration to deeper parts of the pond.
Demethylation and primary productivityVegetative productivity at the sites is likely important to consider when evaluating the differences between the findings of this study and others which found beaver ponds to be methylmercury production hotspots. While I don't know the age of the ponds, my field observations indicate that they are well-established, and have well developed marsh vegetation and algal communities. Increased primary productivity from this vegetation community is a likely source of oxygen which helps create conditions that support demethylation. Evidence of the importance of the vegetation community may be inferred given the importance of pH on methylmercury changes (Figure 17), as primary productivity increases water pH. A chlorophyll A analysis will help to further my understanding of the relationship between primary productivity and methylmercury transformations in the pond.
ConclusionSeasonal changes to flow rate through the ponds drives water chemistry transformations in the beaver ponds. Overall, beaver ponds in the Taiga Plains are not acting as methylmercury hotspots, rather, they are decreasing the methylmercury concentration of water delivered to downstream ecosystems. This is likely due to long residence times which allow for more chemical transformations to occur in a smaller section of the flow path, and suitable redox conditions for demethylation. These conditions which are likely influenced by vegetation communities create the opportunity for demethylation to occur from microbial breakdown, and solar energy breakdown.
|
Figure 17. Photos of site vegetation. Algal and emergent vegetation communities suggest high productivity in, and directly adjacent to the beaver pond.
|